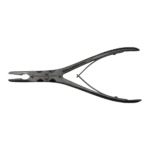
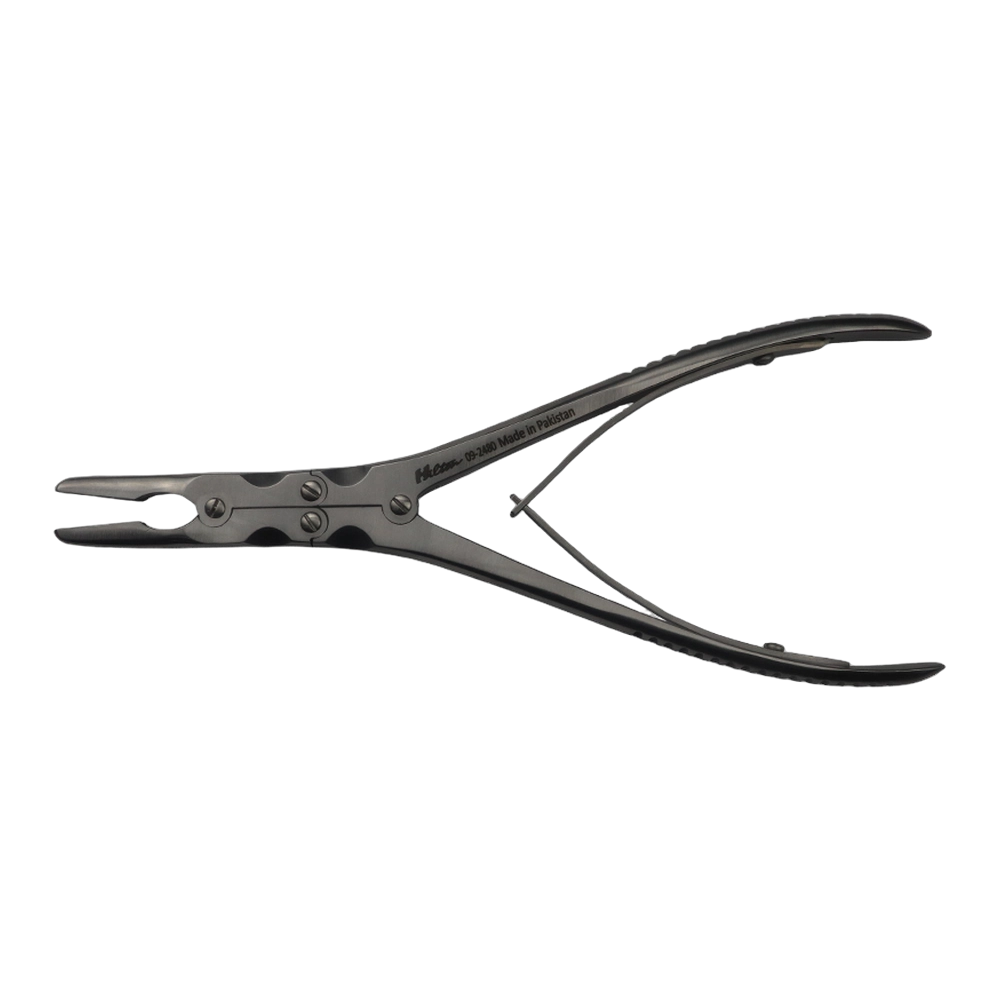
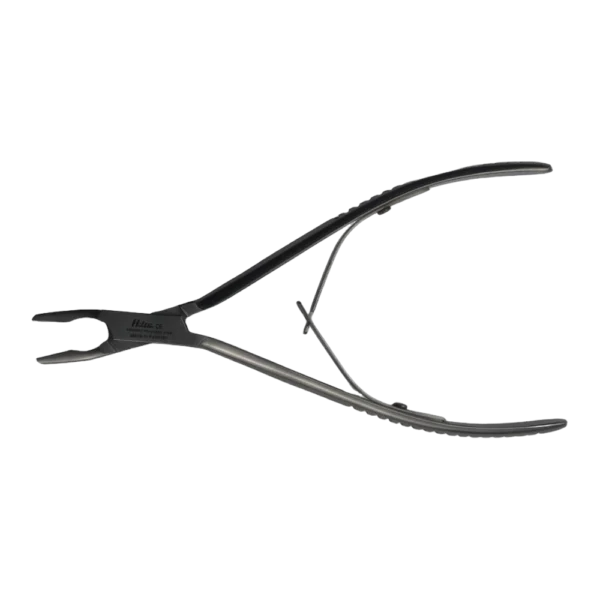
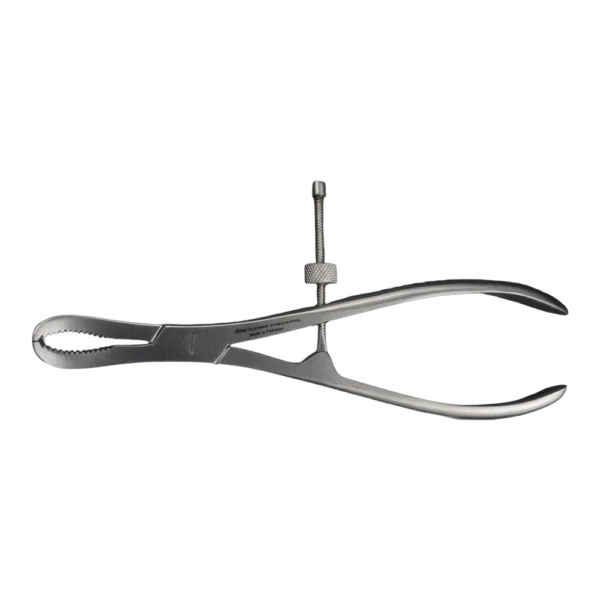
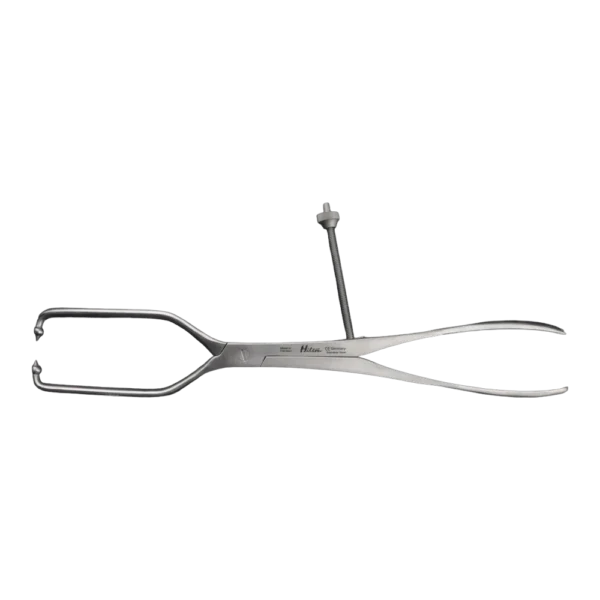
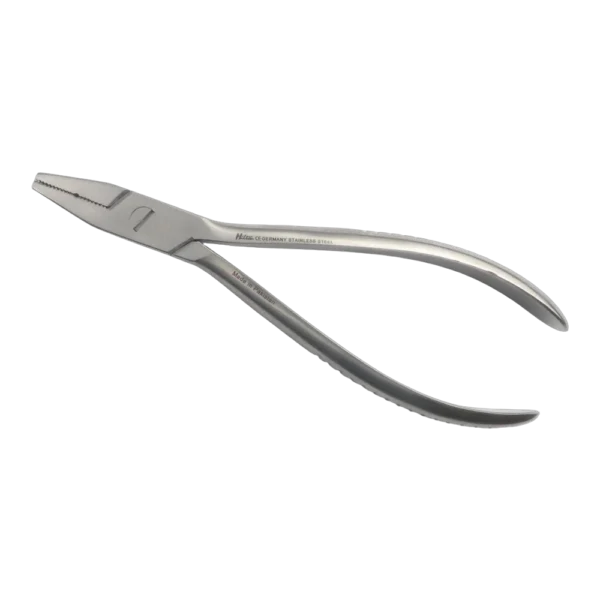
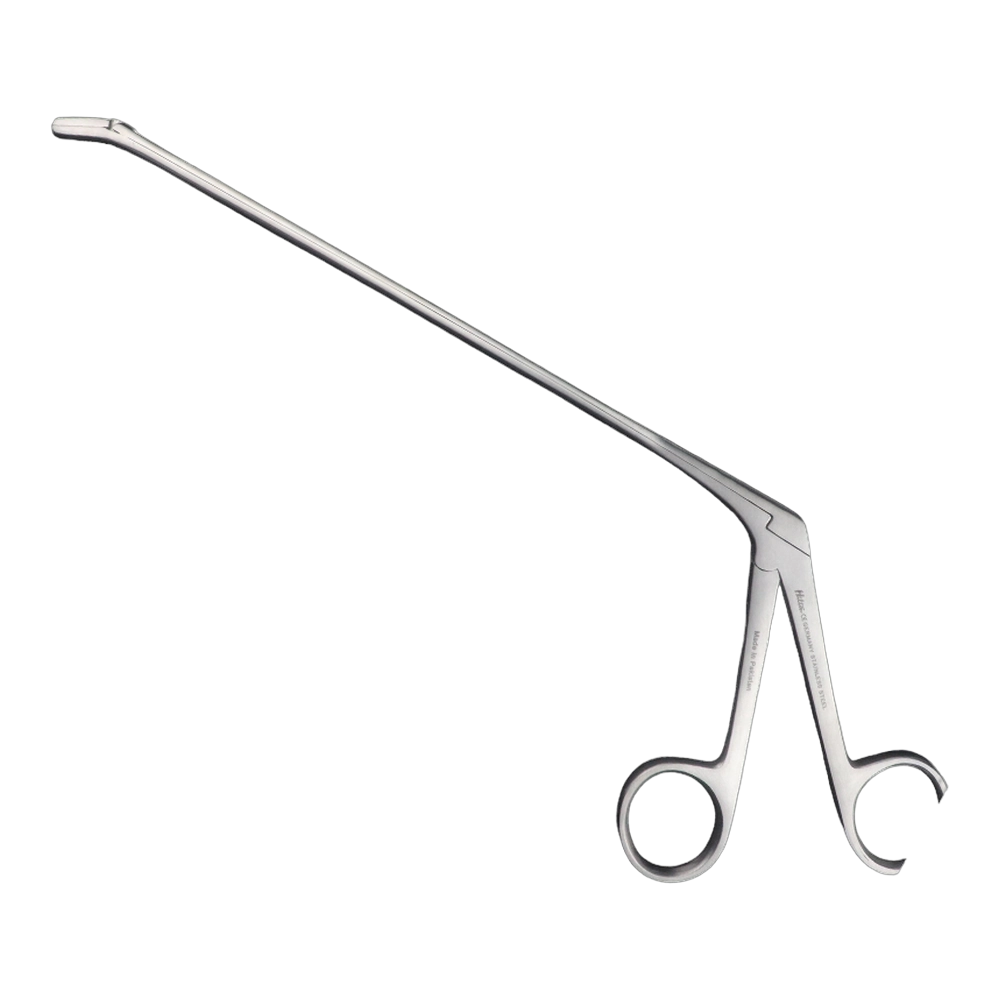
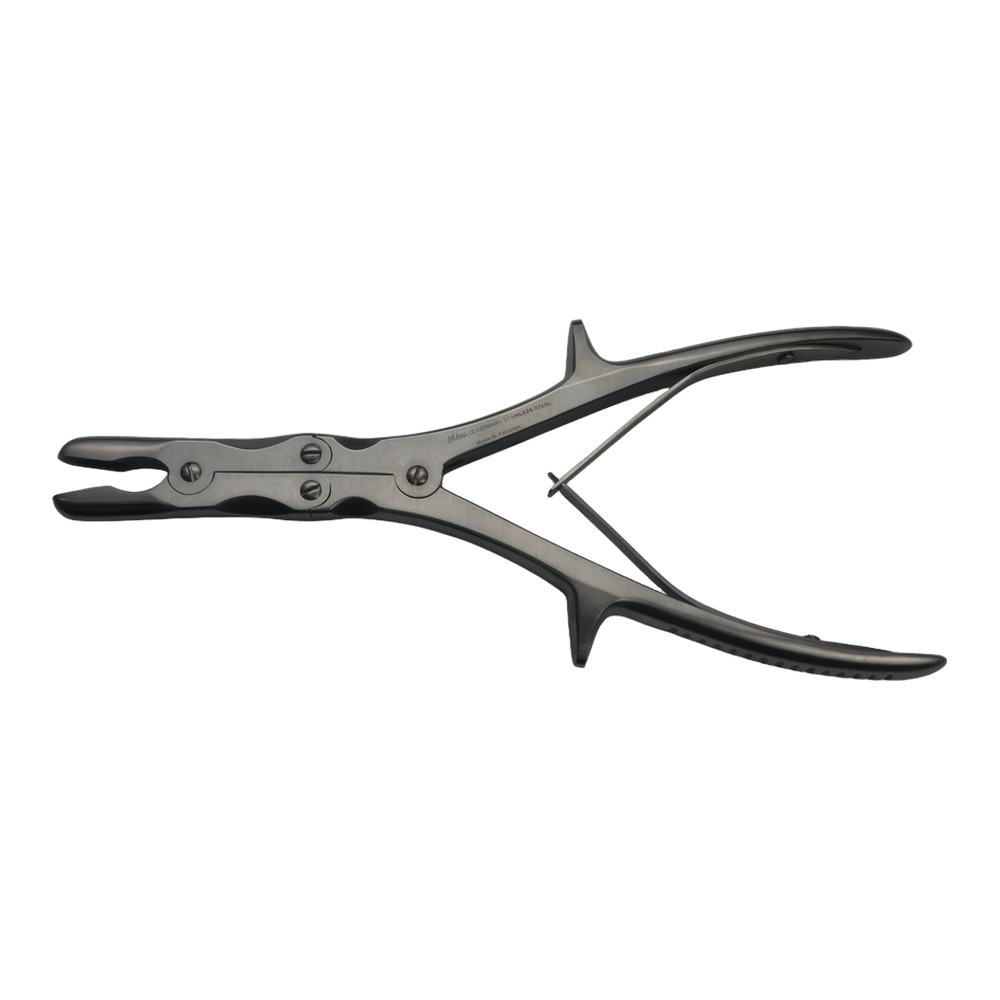
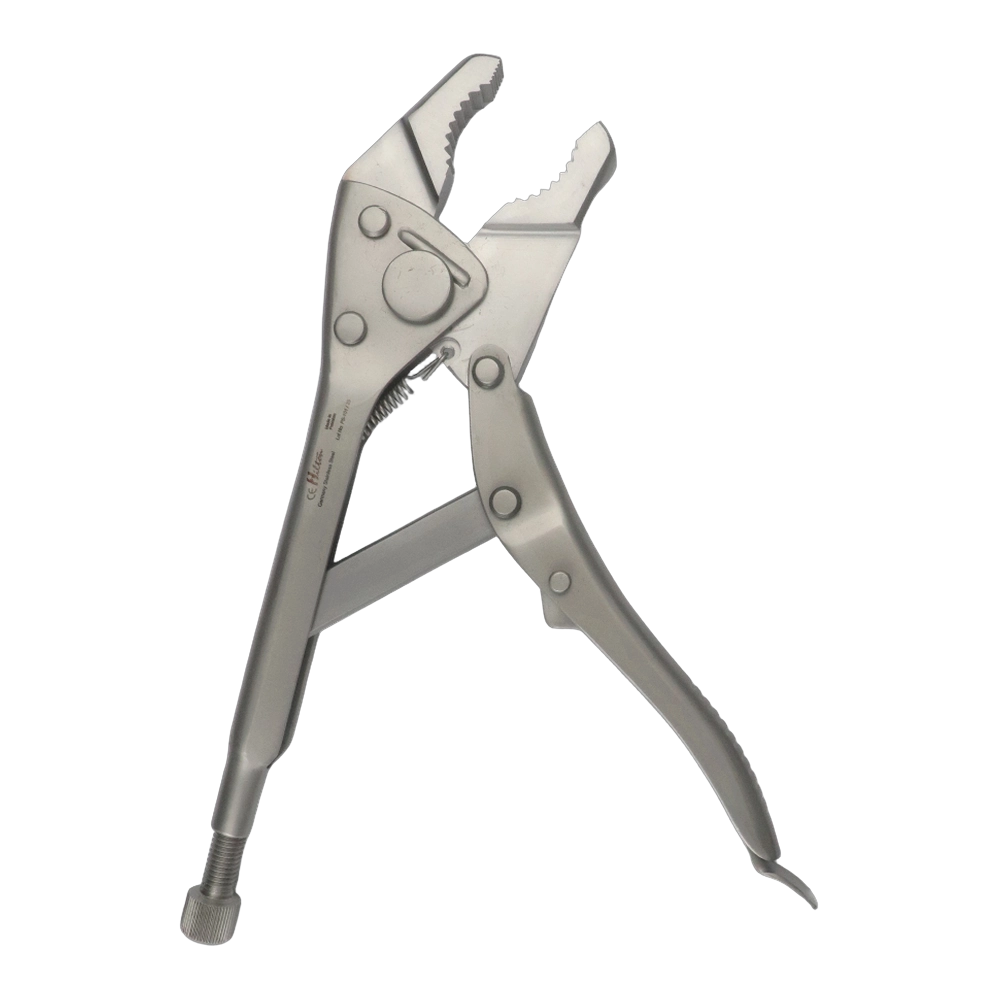
Bone Rongeur Double action 19 cm STR
Provides controlled bone removal and contouring to keep the field clear during exposure. Tactile feedback aids intraoperative judgment and limits instrument changes.
2,860 EGP
more 2 products
6 for the price of 3
* The information on this page is a summary and is not intended to cover all available information about this medication. It does not cover all possible uses, directions, precautions, drug interactions or adverse effects and is not a substitute for the expertise and judgement of your healthcare professional.
Technical Specifications of Bone Rongeur Double action 19 cm STR
| Specification | Description |
|---|---|
| Speciality | Orthopedic |
| Model Variant | 19 cm |
| Material | STAINLESS STEEL |
| Sterilization Method | AUTOCLAVABLE |
| Finish | Matte / Satin Finish |
| Length (mm) | 19 cm |
| Packaging Unit | Single Piece |
| Handle Type | FLAT GRIP |
| Tip Type | STR |
| Brand | Hilton brand |
| Country of Origin | Pakistan |
| SKU | ORT-6003-FOR |
Configured for orthopedic use, this double-action rongeur emphasizes controlled procedural handling with a 19 cm length and flat-grip handles. Constructed from stainless steel with a matte/satin finish, it is compatible with autoclave sterilization for routine reprocessing. Supplied as a single piece with STR tip type under the Hilton brand, manufactured in Pakistan.
Surgical Applications of Bone Rongeur Double action 19 cm STR
Bone Rongeur Double action 19 cm STR enables incremental removal of small bone to support exposure, access preparation, and dimensional adjustment of osseous surfaces. The straight, double-action configuration supports shaping bony edges and graft material for sizing alignment and intended fit within prepared sites.
Common Applications
- Incremental removal of small bone to improve exposure during orthopedic procedures
- Shaping and smoothing bony edges for site preparation
- Contouring and sizing bone graft material for proper fit
- Creating or enlarging cortical windows with controlled bites
- Debridement of loose osseous fragments around fixation or implant sites